Antibiotic Resistance: A Real Threat, Not Doom
Summary
Antibiotic resistance can make everyday infections harder to treat, but this discussion emphasizes it is a serious, manageable threat rather than an imminent end-of-humanity scenario. Resistance happens when bacteria mutate or acquire genes that let them inactivate antibiotics or pump drugs out. A key theme is antibiotic stewardship: avoid antibiotics for viral illness, use the shortest effective course, and do not pressure clinicians for prescriptions. The conversation also highlights real-world examples like MRSA and drug-resistant gram-negative bacteria (including E. coli), plus environmental and agricultural contributors. Practical steps can reduce risk while preserving these life-saving medicines.
🎯 Key Takeaways
- ✓Antibiotic resistance is driven by bacterial mutation or gene sharing, allowing bacteria to break down drugs or pump them out.
- ✓The concern is a gradual slide toward a “pre-antibiotic era,” but the expert frames it as not imminent if antibiotics are used wisely.
- ✓Stewardship means avoiding antibiotics for viral illness and using the shortest effective duration, with modern evidence supporting shorter courses for some infections.
- ✓Drug-resistant bacteria discussed include MRSA and resistant gram-negative organisms such as E. coli, which can also appear in food contamination events.
- ✓Individual choices matter, including not demanding antibiotics, asking about shortest appropriate courses, and disposing of medications properly instead of flushing.
Antibiotics are not just “infection medicines.” They quietly support modern life, including safer surgeries, cancer care, organ transplants, and even routine treatment of common infections.
When antibiotics stop working well, the effects ripple outward. A skin cut that becomes infected can become harder to treat. A urinary tract infection can linger or recur. A hospital stay can become riskier.
This conversation takes a balanced, scientific view: antibiotic resistance is a genuine and growing threat, but it is not framed as an immediate, inevitable extinction event. The key idea is stewardship, meaning protecting antibiotic effectiveness through smarter use.
Did you know? Antibiotic-resistant bacterial infections were associated with about 1.27 million deaths globally in 2019, with resistance contributing to even more deaths overall. That estimate comes from a major analysis in The Lancet reported by the World Health OrganizationTrusted Source.
Why antibiotic resistance matters to your whole health
The fear behind antibiotic resistance is easy to understand. If bacteria learn to evade the drugs we rely on, infections become harder to clear.
The discussion highlights a historical contrast that still shapes medicine today. Before widespread antibiotics (often described as the pre-antibiotic era), infectious diseases were a dominant cause of death. After penicillin and other antibiotics became available, many infections that once killed people became treatable.
The concern is not only about rare “superbugs.” It is about the gradual erosion of a safety net that protects everyday wellbeing. When antibiotics become less reliable, the medical system has fewer easy options, and people can face longer illness, more complications, and more spread within communities.
One important nuance in this perspective is timing.
The risk is described as real, but not imminent, especially if antibiotics are used appropriately and conserved as a shared resource.
Important: Antibiotic resistance is a population-level problem, but the consequences can be very personal. If you have an infection that is not responding to initial treatment, it is a reason to follow up promptly, not a reason to self-adjust doses or share leftover antibiotics.
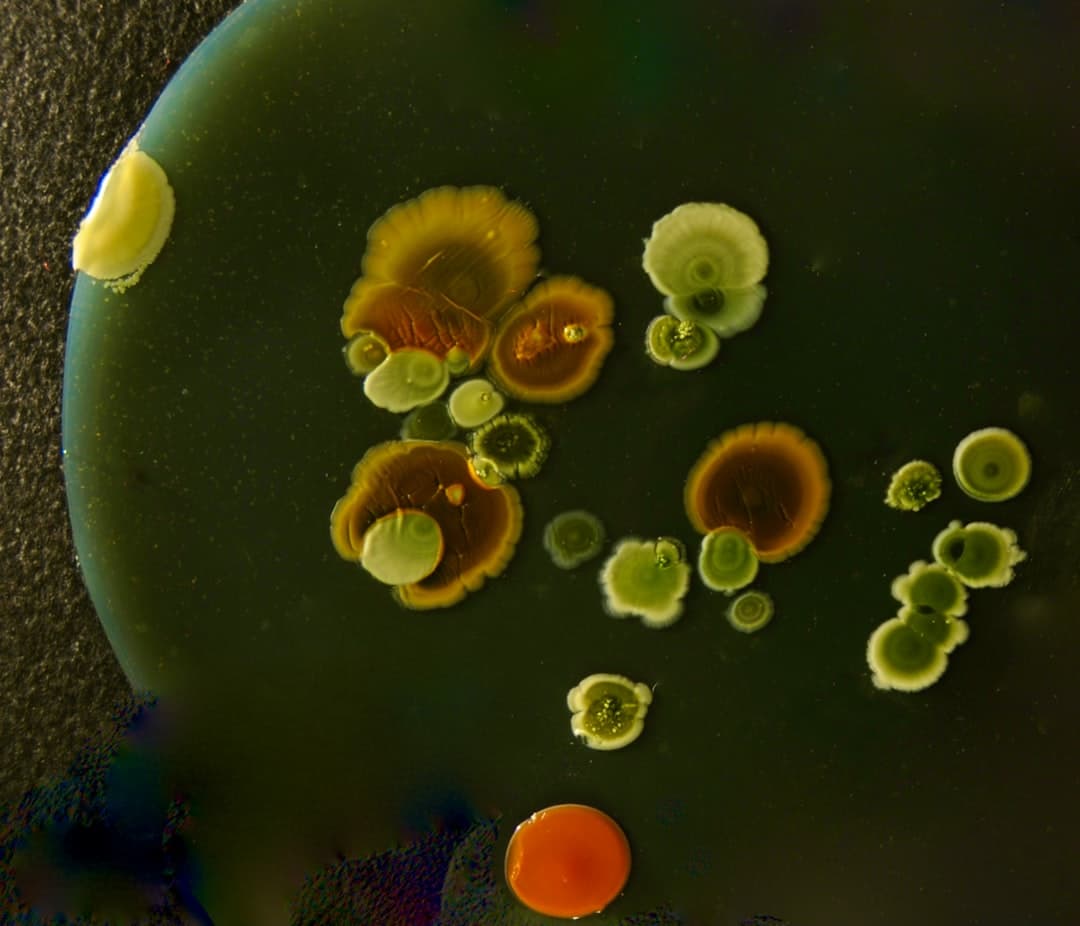
What bacterial resistance is, and how bacteria pull it off
Antibiotic resistance is not your body becoming “used to” a medication.
It is bacteria changing in ways that let them survive exposure to antibiotics.
This framing matters because it shifts the focus from willpower and blame to biology and systems. Bacteria can evolve quickly. They can also share genetic material, which means one bacterium can acquire resistance traits from another.
The main mechanisms discussed
The discussion focuses on two broad routes: mutation and gene acquisition.
From there, bacteria can use several strategies:
The practical consequence is straightforward. A medication that reliably worked for a particular infection in the past may no longer work today.
That is why a person can take an antibiotic and still feel sick, or see symptoms return, even when they did everything “right.” It does not automatically mean the person failed, it may mean the bacteria have changed.
Testing resistance: how clinicians learn what will work
When resistance is suspected, medicine has tools to investigate.
The discussion describes two major approaches.
First, labs can grow the bacteria and expose it to different antibiotics to see what inhibits growth. This is the core idea behind susceptibility testing.
Second, labs can sometimes sequence genes to identify resistance mechanisms. Gene-based testing can help explain why a drug failed, and can guide selection of a more effective alternative.
This is one reason clinicians may ask for cultures, urine samples, wound swabs, or blood cultures in certain situations. It can feel like a delay when you just want relief, but identifying the organism can prevent trial-and-error prescribing.
What the research shows: Surveillance and lab-guided therapy are central strategies in national and global action plans. Public health agencies emphasize that tracking resistance and using diagnostics helps reduce unnecessary antibiotic exposure and improves targeting. See the WHO fact sheet on antimicrobial resistanceTrusted Source.
Stewardship in real life: fewer antibiotics, shorter courses
Stewardship is the backbone of this conversation.
The idea is not “never use antibiotics.” The argument is that antibiotics are among the most life-saving medical technologies, but they should be used carefully, and only when they are likely to help.
Two stewardship themes stand out.
1) Do not use antibiotics for viral infections
Antibiotics do not treat viruses.
This point is emphasized because it is a common conflict in clinics, especially when a child has fever, cough, or flu-like symptoms and caregivers understandably want a tangible treatment.
Using antibiotics for viral illness can still cause harm. The discussion notes consequences for the gut microbiome and potentially for immune balance. More broadly, unnecessary use increases selective pressure that encourages resistant bacteria to persist and spread.
Public health guidance aligns with this. The CDCTrusted Source emphasizes that antibiotics should not be used for colds, flu, many sore throats, and most bronchitis cases, because these are often viral.
Pro Tip: If you are told an illness is likely viral, consider asking, “What symptoms would make you worry about a bacterial infection later?” This keeps the plan concrete without defaulting to an antibiotic.
2) Use the right duration, often shorter than in the past
A distinctive, practical detail in this discussion is how much antibiotic durations have changed.
An example given is uncomplicated pneumonia, described as historically treated for about 7 to 10 days, while more recent practice may use around 3 days in selected uncomplicated cases.
The key insight is not that the antibiotics are “stronger.” It is that evidence has shown the infection can be eradicated sooner in many situations, and longer exposure can add downsides without adding benefit.
Another everyday example is urinary tract infections, where older habits often involved longer courses, while modern approaches may use much shorter regimens, sometimes even a very short course in appropriate situations.
It is still important to follow the plan you are prescribed.
Shorter courses are a clinician decision based on diagnosis, severity, patient factors, and local guidance. If you stop early without guidance, you may undertreat an infection or relapse, and you may still contribute to resistance by exposing bacteria to sub-lethal drug levels.
A practical script for patients
Here is a stewardship-oriented way to communicate without second-guessing your clinician:
»MORE: Consider looking up your local public health “antibiotic awareness” resources, many regions publish one-page guides on when antibiotics help and when they do not. The CDC’s Be Antibiotics AwareTrusted Source pages are a good starting point.
The bacteria that keep showing up: MRSA and gram-negatives
Some resistant bacteria have become household names.
Two categories are highlighted in the discussion because they represent different challenges.
MRSA
MRSA stands for methicillin-resistant Staphylococcus aureus.
This bacterium can cause skin and soft tissue infections, and in more serious cases, invasive infections. Resistance means some commonly used antibiotics may not work, requiring different choices.
The CDC’s MRSA informationTrusted Source describes how MRSA can spread in healthcare settings and in the community, and emphasizes hygiene and appropriate care for wounds.
Gram-negative bacteria like E. coli
The discussion also points to gram-negative organisms, including E. coli and related bacteria, as a major resistance concern.
Gram-negative bacteria have structural features that can make them harder to treat, and they can accumulate multiple resistance mechanisms.
This category also connects to news events people recognize, such as food contamination outbreaks. Many outbreaks involve strains that are not highly resistant, but the broader point stands: common bacteria can become difficult to treat when resistance rises.
A useful takeaway is that resistance is not confined to exotic pathogens. It can involve microbes that live in and around us all the time.
Expert Q&A
Q: If antibiotics saved so many lives, why not use them “just in case”?
A: The “just in case” approach can backfire. If the illness is viral, antibiotics will not shorten the course, but they can still cause side effects and disrupt the gut microbiome. They also increase selective pressure that helps resistant bacteria survive and spread.
This perspective treats antibiotic effectiveness as a shared resource. The goal is to preserve these medicines so they still work when a bacterial infection truly needs them.
Dr. Tom Warren, Infectious Disease Physician (as featured in the video)
Beyond the clinic: agriculture, environment, and global access
Antibiotic resistance is not only about what happens in a doctor’s office.
The discussion raises three broader drivers that can shape what bacteria we encounter.
Antibiotics in animal agriculture
Antibiotic use in livestock is described as a significant issue globally.
The concern is not a single pathway, it is the overall selection pressure that encourages resistant organisms to develop and circulate through food systems, waste, and the environment.
The WHOTrusted Source has discussed how antimicrobial use in food-producing animals can contribute to resistance and why reducing unnecessary use matters.
Environmental exposure, including water sources
A striking point is that resistant bacteria can be detected in the environment, including water sources, because antibiotics and resistant organisms can be excreted and dispersed.
This does not mean everyday water exposure will automatically cause infection. It means resistance is ecological, not just clinical.
Medication disposal and “down the toilet” habits
The conversation makes a practical note: do not flush medications.
Instead, use pharmacy take-back programs or community disposal sites.
In the United States, the FDA guidance on drug disposalTrusted Source explains take-back options and when flushing is recommended only for a small list of high-risk drugs.
Over-the-counter access in some countries
Another global driver discussed is that in some regions antibiotics can be purchased without a prescription.
That increases the chance that antibiotics are used for viral illnesses or used incorrectly (wrong drug, dose, or duration). The result is more resistance pressure, affecting not only individuals but also communities and international travel networks.
Can antibiotic resistance cause the next pandemic?
This discussion separates two ideas that often get blended online: antibiotic resistance as a slow-moving crisis, and pandemics as fast-moving events.
The view presented is that a pandemic like the recent global event is more likely to be viral than bacterial.
The reason is transmission dynamics. Many bacterial infections spread person-to-person more slowly and often require closer contact or specific routes. Viruses, in general, can have higher infectivity and faster spread.
That said, bacterial pandemics have occurred historically, with plague (caused by Yersinia pestis) cited as a major example.
So the key distinction is not “bacteria cannot cause pandemics.” It is that the conditions for rapid global spread are often more favorable for viruses.
Antibiotic resistance still matters in outbreaks and emergencies. Secondary bacterial infections, hospital-acquired infections, and invasive procedures during crises can all become harder to manage if resistance is widespread.
Expert Q&A
Q: What can an average person do that actually makes a difference?
A: The most actionable steps are behavioral. Avoid pressuring clinicians for antibiotics when symptoms suggest a viral illness, and ask about the shortest appropriate duration when antibiotics are needed. Also avoid sharing leftover antibiotics and dispose of unused medications through take-back programs.
These steps reduce unnecessary antibiotic exposure, which helps slow the selection of resistant bacteria in the community.
Dr. Tom Warren, Infectious Disease Physician (as featured in the video)
Key Takeaways
Frequently Asked Questions
- What is antibiotic resistance in simple terms?
- Antibiotic resistance means bacteria have changed so that an antibiotic no longer kills them or stops their growth. It can happen through mutations or by bacteria acquiring resistance genes from other bacteria.
- Why won’t antibiotics help my child’s viral cold or flu?
- Antibiotics target bacteria, not viruses, so they usually will not shorten viral illnesses. Unnecessary antibiotics can still cause side effects and can contribute to resistance over time.
- Is it true that shorter antibiotic courses can be enough?
- For some common infections, research and updated practice have supported shorter courses than were used decades ago. The right length depends on the infection and the person, so it is worth asking your clinician what duration is appropriate for you.
- Which bacteria are commonly associated with resistance?
- Examples often discussed include MRSA (methicillin-resistant Staphylococcus aureus) and resistant gram-negative bacteria such as E. coli. These organisms can be harder to treat because fewer antibiotics remain effective.
- Can antibiotic resistance cause a pandemic like COVID-19?
- Large, fast-moving pandemics are more commonly caused by viruses because they often spread more easily between people. Bacterial pandemics have happened historically, but the discussion emphasizes viruses as the more likely driver of rapid global spread.
Get Evidence-Based Health Tips
Join readers getting weekly insights on health, nutrition, and wellness. No spam, ever.
No spam. Unsubscribe anytime.